- Blog
- Delete photo google search
- 4 pack deadbolt locks
- Free download garageband windows 10
- Plain zoom backgrounds free
- Computer spreadsheets for monthly expenses
- Solitaire classic free online games
- Checklist for moving a small business
- Erie county real estate transactions january 28
- Lyfe jennings 268 192 download zip
- Download the sims 4 on chromebook
- Kink compatibility checklist
- Moca test score 2019
- Periodic table magnesium sulfate molar mass
- Nurse student resume template baylor
- X ray cervical spine tumor
- Free games spider solitaire
- Download gta 5 on ps4 free
- List of dog commands and hand signals
- Claude monet impression sunrise medium
- Generic cover letter examples
- Xplane 11 pro use key
- Outdoor christmas lights projectors onto house
- Starcraft remastered zoom out
- Whatsapp dark mode apk download latest version
- Kink checklist scat
- Minions beach butt pinterest
- Manycam download old version
- Dordogne river region of france map
- Guitar hero live wii u dongle
- Live anime wallpapers pc
- Demonstar download full version
- Serial number adobe premiere pro cs6
- Monthly living expenses spreadsheet
- Krunker transparent scope image
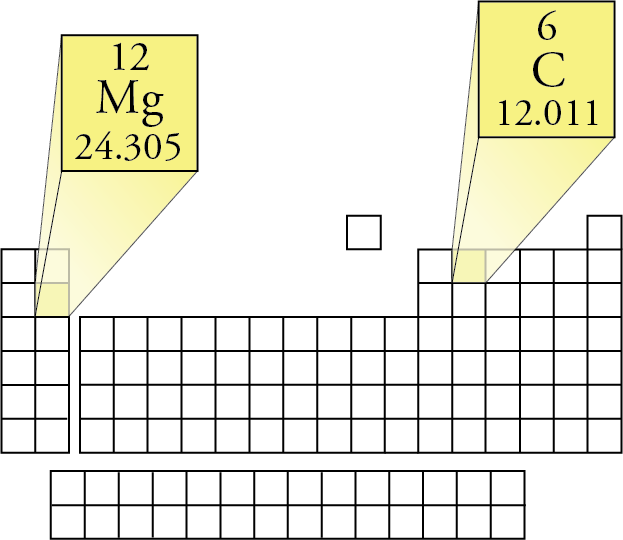
CO 2 has one carbon atom and two oxygen atoms.Oxygen (O) has an atomic mass of about 16.00 amu.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.You can see that The molar mass of Sodium is 22.990 g/mol. So let’s look at the molar mass of Sodium, Sulfur and Oxygen from the above periodic table. Now in Na2SO4, there are 2 Sodium atoms, 1 Sulfur atom and 4 Oxygen atoms. Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. You can see the molar mass value of all the atoms from this periodic table.

The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find atomic masses: look up the atomic masses of each element present in the compound.For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound.One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) Mole is a standard scientific unit for measuring large quantities of very small entities such as atoms and molecules.Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol.You can see that The molar mass of Copper is 63.546 g/mol. So let’s look at the molar mass of Magnesium and Bromine from the above periodic table. Now in MgBr2, there is 1 Magnesium atom and 2 Bromine atoms. (1 u is equal to 1/12 the mass of one atom of carbon-12) You can see the molar mass value of all the atoms from this periodic table. Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u).To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations: Use a small funnel to pour 25 cm 3 of dilute hydrochloric acid into the burette, followed carefully by 25 cm 3 of water. The mass should lie between 0.020 and 0.040 g. Definitions Question Solutions Practice Chemistry Quizzes Info Chemical Formulas Point Group Symmetry Fun Chemistry Jokes Words from Elements Chat Forums ENEnglish Login MgSO4 (Magnesium Sulfate) Molar Mass Magnesium Sulfate 24. Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Weigh accurately, to the nearest 0.001 g, a length of magnesium ribbon, approximately 34 cm long. Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Tos, Bz, TMS, tBu, Bzl, Bn, DmgĮxamples of molar mass computations: NaCl, Ca(OH)2, K4, CuSO4*5H2O, nitric acid, potassium permanganate, ethanol, fructose, caffeine, water.You can also use this molarity calculator to find the mass concentration or molar mass. Substitute the known values to calculate the molarity: molarity 5 / (1.2 × 36.46) 0.114 mol/l 0.114 M. As mass/volume molarity × molar mass, then mass / (volume × molar mass) molarity. Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. Convert the expressions above to obtain a molarity formula. 109535).Magnesium sulfite appears as a white crystalline solid.Ĭomputing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'. Before placing in Container D, check the pH with pH-Universal indicator strips (Item No. Now in MgSO4, there is 1 Magnesium atom, 1 Sulfur atom and 4 Oxygen atoms. Home » Chem » Compound Search Magnesium Sulfate. You can see the molar mass value of all the atoms from this periodic table. Neutral solutions of the these salts: Container D. Magnesium Sulfate MgSO4 Molecular Weight, molar mass converter. The molar mass of MgSO4 (Magnesium sulfate) is: 120.361 grams/mol. Atomic weight of Magnesium (Mg): 24.305 (Ref: Jlab-ele012) Atomic weight of Sulfur (S): 32.066 (Ref: Jlab-ele016) Atomic weight of Oxygen (O): 15.9994 (Ref: Jlab-ele008) Step 3: Calculate the molecular weight of Magnesium sulfate by adding the total weight of all atoms. The periodic table lists the atomic mass of carbon as 12.011 amu. Step 2: Find out the atomic weights of each atom (from the periodic table). The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. Bitter salt, epsom salt, sulfuric acid magnesium salt heptahydrateġ4 Inorganic salts: Container I. In each case, the number of grams in 1 mol is the same as the number of atomic mass units that describe the atomic mass, the molecular mass, or the formula mass, respectively.
